SUPPORTED BY COMPARATIVE
CLINICAL EVIDENCE
Bosaya™ has achieved all benchmarks for biosimilarity in efficacy and safety to the reference denosumab.1-3
Totality of evidence for BOSAYA is based on similarity in analytical and clinical data, with extrapolation to Prolia® indications.1-3
EFFICACY
STUDY DESIGN1
The DEVOTE study was a randomized, double-blind, multicenter, parallel-arm, Phase 3 study that compared the efficacy, pharmacodynamics, safety, and immunogenicity of BOSAYA and Prolia in postmenopausal women with osteoporosis.
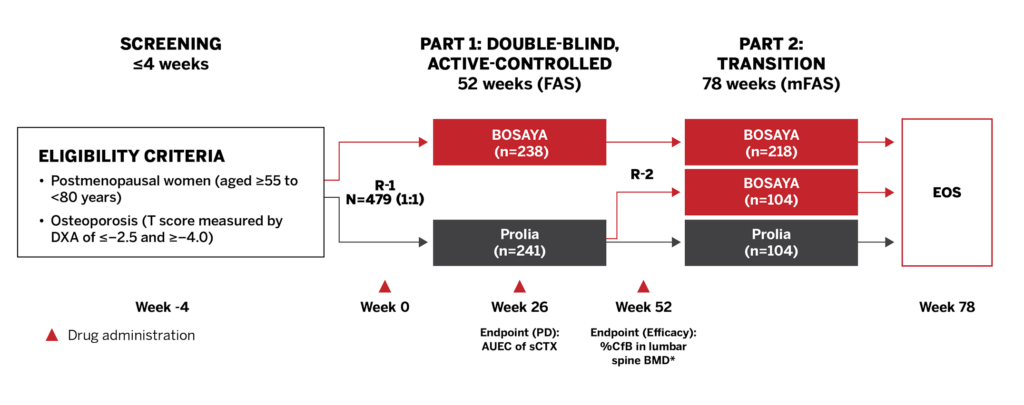
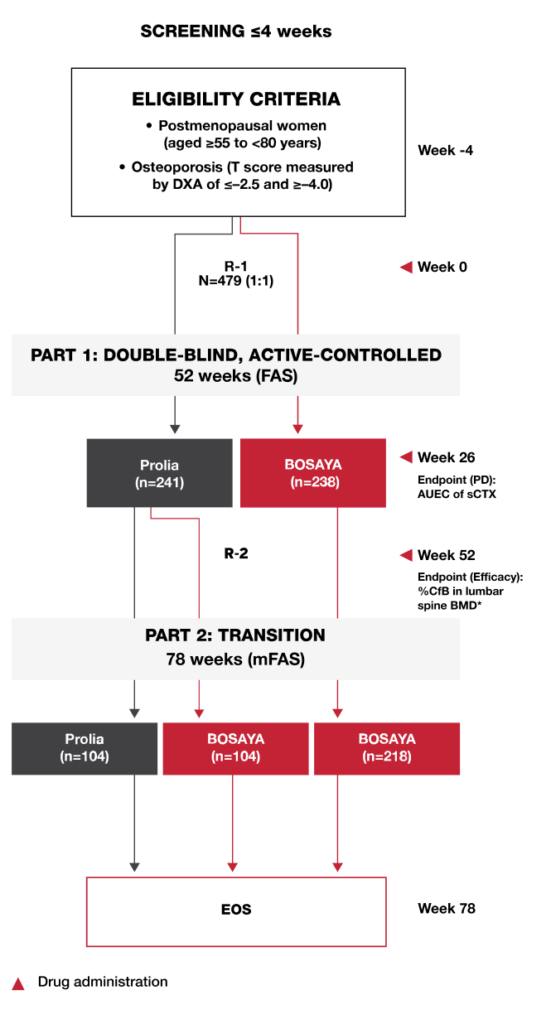
Study Endpoints1
- The co-primary endpoints were %CfB in the lumbar spine BMD by DXA at Week 52 and AUEC of sCTX at Week 26
- Key secondary endpoints included %CfB in femoral neck BMD and total hip BMD at Weeks 26, 52, and 78
- Immunogenicity was compared based on the incidence of ADAs and NAbs
- Safety, efficacy, and immunogenicity were compared after a single switch at Week 52 up to Week 78
Baseline Characteristics1
-
- The key baseline demographic characteristics were comparable between the treatment groups with respect to age, race, height, and weight
*As measured by DXA.
%CfB=percent change from baseline; ADAs=anti-drug antibodies; AUEC=area under the effect curve; BMD=bone mineral density; DXA=dual-energy x-ray absorptiometry; EOS=end of study; FAS=full analysis set; mFAS=modified full analysis set; NAbs=neutralizing antibodies; PD=pharmacodynamic; R-1=randomization 1 on Day 1; R-2=randomization 2 at Week 52; sCTX=serum C-terminal telopeptide of Type 1 collagen.
LUMBAR SPINE BMD1
Percent change from baseline in lumbar spine BMD at Week 521
- BOSAYA demonstrated equivalence to the Prolia product in lumbar spine BMD at Week 52 (primary endpoint)
- The 90% CI of the difference in percent change from baseline in lumbar spine BMD (0.601%) was within the prespecified equivalence margin of -1.45%, 1.45%
Percent change from baseline in lumbar spine BMD at Week 781
- Switching at Week 52 and continuing on BOSAYA maintained comparable clinical efficacy up to Week 78
- Similar results were seen for hip BMD and femoral neck BMD
BMD=bone mineral density; CI=confidence interval.
ABSOLUTE CONCENTRATION OF sCTX1
- BOSAYA showed similar biological effect on bone turnover as demonstrated by a comparable decrease in sCTX levels at Week 26
- The 95% CI of the geometric mean ratio for AUEC (104.12%) was within the prespecified equivalence margin of 80.0%, 125.0%
AUEC=area under the effect curve; CI=confidence interval; sCTX=serum C-terminal telopeptide of Type 1 collagen.
SAFETY
SIMILAR SAFETY AND IMMUNOGENICITY PROFILE1
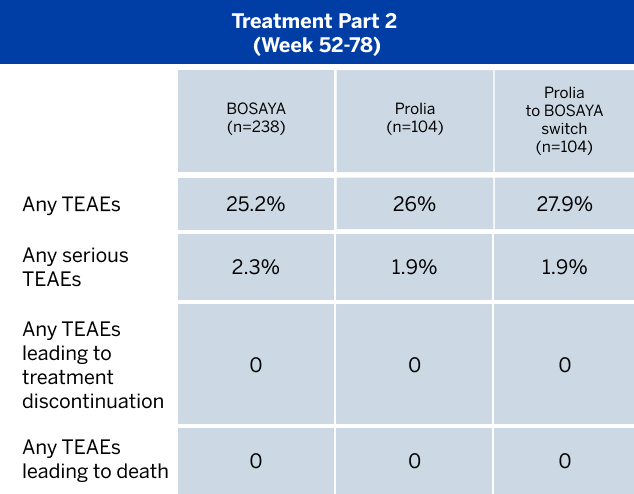
Safety and immunogenicity were found to be similar between the treatment groups at Week 52, and after switching at Week 52 up to Week 78.
OVERALL INCIDENCE OF TREATMENT-EMERGENT ADVERSE EVENTS (TEAEs)1
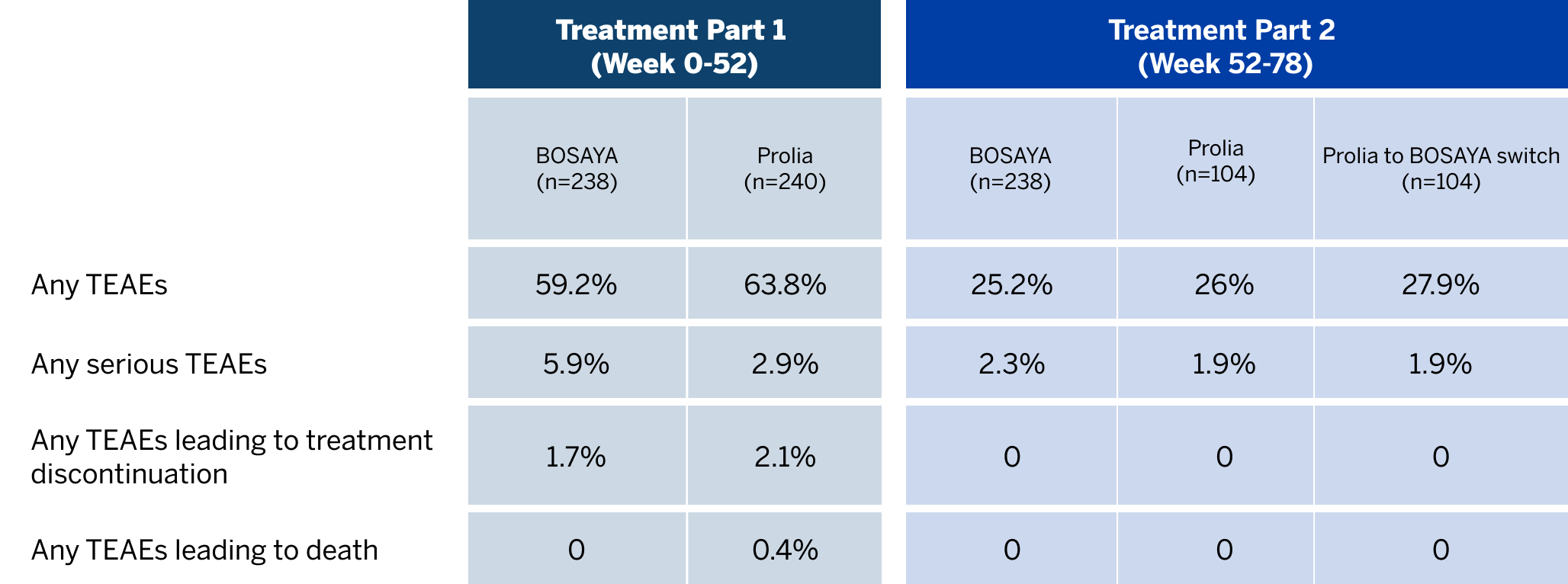
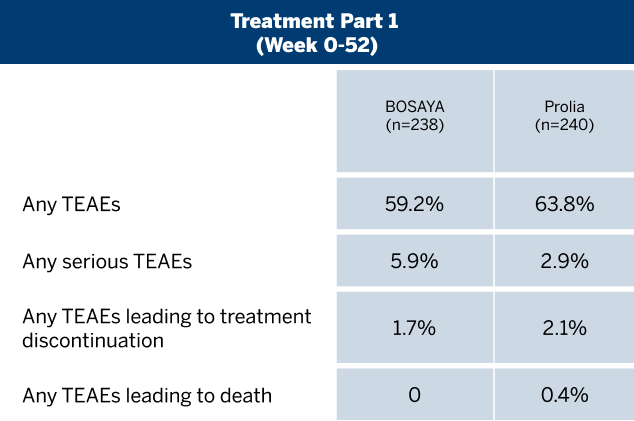
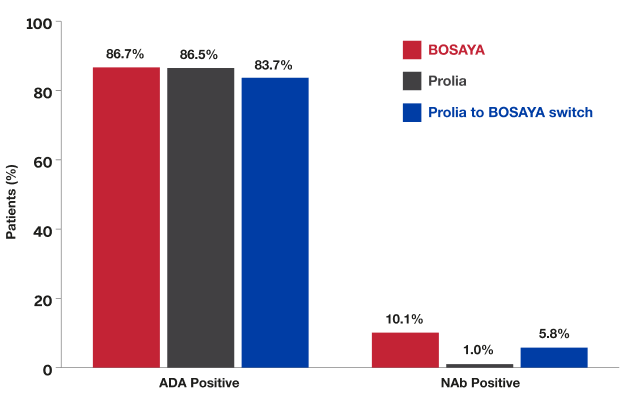
Bosaya immunogenicity compared to prolia1
- Data shown refer to the same 78-week, double-blind trial as the efficacy data; please see the study design under the efficacy section
- The proportion of patients positive for ADAs was high and similar between treatment groups
- The proportion of patients with NAbs was very low and similar between both groups
ADAs=anti-drug antibodies; NAbs=neutralizing antibodies.
REFERENCES
1. Data on file. Biocon Biologics; 2025. 2. BOSAYA. Prescribing information. Biocon Biologics Inc; 2025. 3. US Food and Drug Administration. Biosimilars basics for patients. Updated August 1, 2024. Accessed October 1, 2025. https://www.fda.gov/drugs/biosimilars/biosimilar-basics-patients
