An interchangeable denosumab biosimilar1
Protect What Moves Them
BosayaTM gives them the strength to keep moving with reduced risk of fractures.1


Deliver Quality Care With Confidence.
BOSAYA is a treatment option as safe and as effective as Prolia®.1
BOSAYA and Prolia share the same1-3:

FDA-approved indications

Dosing schedule
Mechanism of action

Expected clinical outcomes
BOSAYA is indicated for the treatment of patients at high risk for fracture with1:
Postmenopausal osteoporosis
Glucocorticoid-induced osteoporosis
Prostate or breast cancer treatment-induced bone loss
Making Movement Between Treatments as Seamless as Possible.
BOSAYA is available for subcutaneous injection in a 60 mg/mL, single-dose prefilled syringe for twice-yearly dosing.1
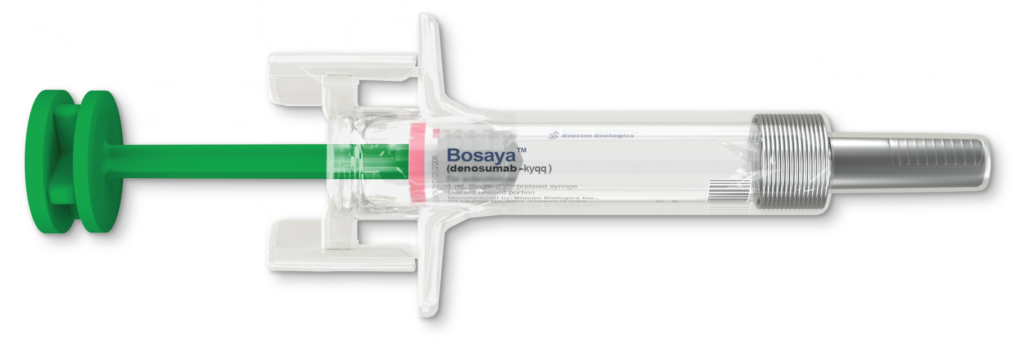
29G Needle
Thinner needle size than Prolia1
Latex Free
Reduces hypersensitivity reactions1
Needle Safety Guard
Prevents needlestick injuries1
See the Power of Proven Results.
Help patients achieve their bone mass density targets with a biosimilar that works as effectively and safely as Prolia.1,2


STRONGER WITH
SUPPORT.
You Prescribe, We Provide
From the moment you prescribe BOSAYA, Biocon Biologics is here to help get your patients started on therapy.
Coding and Billing
The Coding Tip Sheet is a quick-reference tool.
The Coding and Billing Guide
provides more detailed information.
PRIOR AUTHORIZATION ASSISTANCE
Obtain assistance with requirements and submission details, and status tracking.
BENEFITS INVESTIGATION
Receive help with patient-specific insurance coverage requirements and patient cost-sharing aspects such as coinsurance and out-of-pocket maximum.
MY BIOCON BIOLOGICS™
Specialists are available to help Monday-Friday 8 AM-8 PM ET by calling 1-833-612-4626.
PATIENT SERVICES
Enroll patients in support including patient benefits investigation and more.
BOSAYA has been assigned the HCPCS
Level II Product Code of Q5161.

Helping Keep Costs in Check.
Biosimilars such as BOSAYA are an effective tool to help offset rising healthcare costs by offering treatment alternatives.
- RISING COSTS
- PROVEN SAVINGS
$2.2B
more in spending for 677,400 Prolia patients on Medicare in 20234
$20.2B
in healthcare savings from biosimilars in 2024 alone5
Over $56 billion saved since biosimilars entered the US market5
Over $56 billion saved since biosimilars entered the US market5
Biocon Biologics is committed to global leadership in biosimilars.
By delivering affordable access to high-quality biosimilars such as BOSAYA around the world, Biocon Biologics aims to help make a meaningful difference for you and your patients.
Learn more about us at BioconBiologicsUS.com
REFERENCES
1. BOSAYA. Prescribing information. Biocon Biologics Inc; 2025. 2. US Food and Drug Administration. Biosimilars: review and approval. Last updated December 13, 2022. Accessed October 24, 2025. https://www.fda.gov/drugs/biosimilars/review-and-approval 3. Prolia. Prescribing information. Amgen Inc; 2025. 4. MedPAC. Prescription drugs. In: A Data Book: Health Care Spending and the Medicare Program, July 2025. Section 10. Accessed November 20, 2025. https://www.medpac.gov/wp-content/uploads/2025/07/July2025_MedPAC_DataBook_Sec10_SEC-1.pdf 5. Association for Accessible Medicines. The U.S. Generic & Biosimilar Medicines Savings Report. September 2025. Accessed November 20, 2025. https://accessiblemeds.org/wp-content/uploads/2025/09/AAM-2025-Generic-Biosimilar-Medicines-Savings-Report-WEB.pdf
